Click chemistry describes a class of chemical reactions that use bio-orthogonal or biologically unique moieties to label and detect a molecule of interest in mild, aqueous conditions. DBCO alkynes can be used to perform click reactions with azide-modified targets without the use of heavy metal catalysis. DBCO reactions are ideal for surface labeling of live cells and also minimize damage to fluorescent proteins like GFP or R-PE.
The biotin DBCO is reactive with azide via a Strain-promoted Azide-Alkyne Click Chemistry reaction (SPAAC). Biotin can be subsequently detected with streptavidin, avidin or NeutrAvidin® biotin-binding protein.
Features
- Efficiency—the click reaction is complete in less than 1 hour;
- Specificity—the reaction between the label and detection tag is selective and specific;
- Stability—the reaction product contains an irreversible, covalent bond;
- Biologically inert—the components of the reaction do not undergo any side reactions.
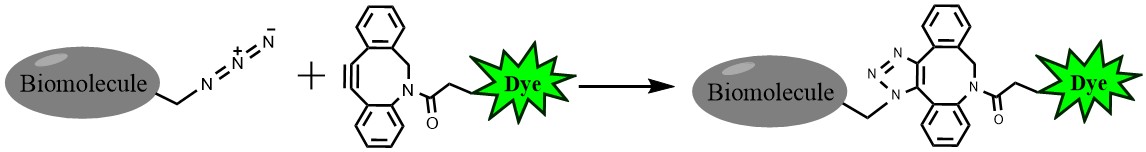
Figure 1. Click chemistry labeling
Specifications
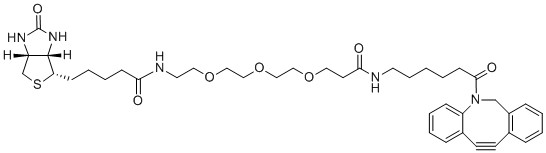
| Label: | Biotin |
|
| Ex/Em: | – | |
| Detection Method: | – | |
| Solubility: | DMSO, DMF | |
| Molecular Weight: | 747.95 | |
| Product Size: | 1 mg | |
| Storage Conditions: | -20 ℃, protect from light | |
| Shipping Condition: | Room Temperature | |
|
Applications Click chemistry labeling |
||

