Click chemistry describes a class of chemical reactions that use bio-orthogonal or biologically unique moieties to label and detect a molecule of interest in mild, aqueous conditions. The click reaction involves a copper-catalyzed triazole formation from an azide and an alkyne. The azide and alkyne moieties can be used interchangeably; either one can be used to tag the molecule of interest, while the other is used for subsequent detection.
Reference
The Andy Fluor™ 647 azide is reactive with terminal alkyne via a copper-catalyzed click reaction that allows the subsequent visualization by fluorescence spectroscopy.
Features
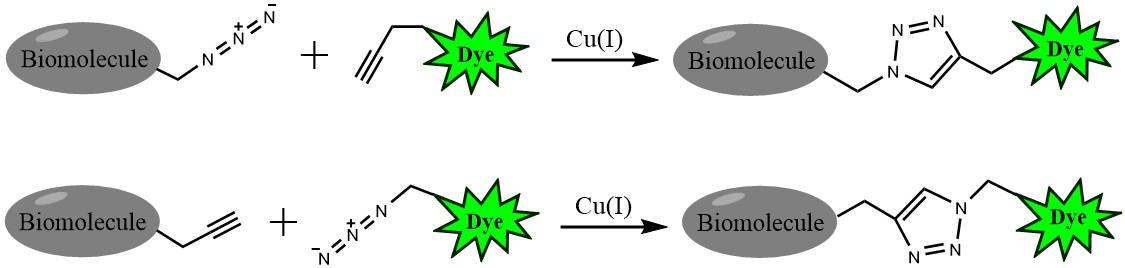
Figure 1. Click chemistry labeling
Specifications
Label:
Andy Fluor™ 647
Ex/Em:
650/666
Detection Method:
Fluorescent
Solubility:
DMSO, DMF
Product Size:
1 µmol
Storage Conditions:
-20 ℃, protect from light
Shipping Condition:
Room Temperature
Applications
Click chemistry labeling
Datasheet (PDF):
C324
MSDS (PDF):
C324
Angew Chem Int Ed Engl (2012) 51:3143-3146
Nucleic Acids Res (2012) 40:e78-e78
Proc Natl Acad Sci U S A (2013) 110:4992-4997
Proc Natl Acad Sci U S A (2007) 104:2614-2619
Angew Chem Int Ed Engl (2009) 48:4030-4033
Related Products
Home » Click Chemistry Tools » Andy Fluor™ 647 Azide
Andy Fluor™ 647 Azide
Introduction
To order
Documents
Dumont A, Malleron A, Awwad M, Dukan S, Vauzeilles B,
Winz ML, Samanta A, Benzinger D, Jäschke A,
Ngo JT, Schuman EM, Tirrell DA,
Hsu TL, Hanson SR, Kishikawa K, Wang SK, Sawa M, Wong CH
Chang PV, Chen X, Smyrniotis C, Xenakis A, Hu T, Bertozzi CR, Wu P
Related products
Contact Us
ABP Biosciences
405 E Gude Dr, STE 214
Rockville, MD 20850
Service Hotline
Tel: 301-658-7993
E-mail: info@abpbio.com
Copyright@ 2026 ABP Biosciences, LLC. All Rights Reserved.

