Click chemistry describes a class of chemical reactions that use bio-orthogonal or biologically unique moieties to label and detect a molecule of interest in mild, aqueous conditions. The click reaction involves a copper-catalyzed triazole formation from an azide and an alkyne. The azide and alkyne moieties can be used interchangeably; either one can be used to tag the molecule of interest, while the other is used for subsequent detection.
Reference
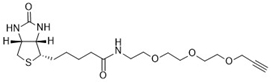
The biotin alkyne is reactive with azide via a copper-catalyzed click reaction. Biotin can be subsequently detected with streptavidin, avidin or NeutrAvidin® biotin-binding protein.
Features
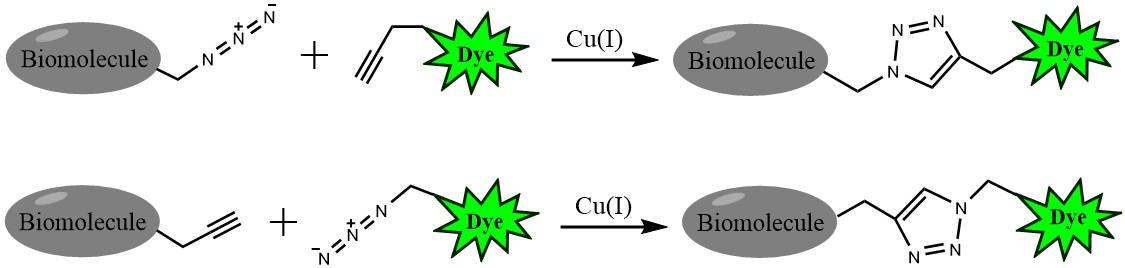
Figure 1. Click chemistry labeling
Specifications
Label:
Biotin
Ex/Em:
–
Detection Method:
–
Solubility:
DMSO, DMF
Molecular Weight:
413.53
Product Size:
5 mg
Storage Conditions:
-20 ℃, protect from light
Shipping Condition:
Room Temperature
Applications
Click chemistry labeling
Datasheet (PDF):
C303
MSDS (PDF):
C303
J Am Chem Soc (2008) 130:11576-11577
J Am Chem Soc (2009) 131:4967-4975
J Biol Chem (2009) 284:21327-21337
Home » Click Chemistry Tools » Biotin Alkyne
Biotin Alkyne
Introduction
To order
Documents
Clark PM, Dweck JF, Mason DE, Hart CR, Buck SB, Peters EC, Agnew BJ, Hsieh-Wilson LC,
Charron G, Zhang MM, Yount JS, Wilson J, Raghavan AS, Shamir E, Hang HC,
Dias WB, Cheung WD, Wang Z, Hart GW,
Contact Us
ABP Biosciences
405 E Gude Dr, STE 214
Rockville, MD 20850
Service Hotline
Tel: 301-658-7993
E-mail: info@abpbio.com
Copyright@ 2026 ABP Biosciences, LLC. All Rights Reserved.

