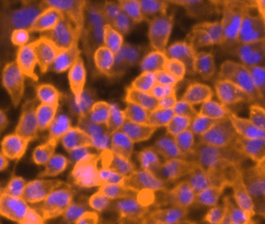
DiI is an orange-red fluorescent, lipophilic carbocyanine and widely used as a long-term tracer for neuronal and other cells. It is weakly fluorescent in water but highly fluorescent when incorporated into membranes. Once applied to cells, the dye diffuses laterally to stain the entire cell.
Reference:
Frequently Asked Questions (FAQs)
I need to look at live cell morphology deformation over the course of a few hours. What sort of membrane dye would be useful for this?
I’m labeling live cells with DiI or DiD lipophilic cyanine dyes. DiI gives a nice even membrane labeling, but DiD is more “spotty”. What can be done?
I want to perform a cell fusion assay, where one cell line is labeled with one color and the other cell line with another color, and combine with a nucleic acid stain. What do you recommend?
Specifications:
Excitation/Emission:
550/565 nm
Shipping Condition:
Ambient
Storage Conditions:
-20ºC, protect from light
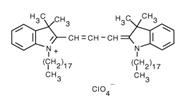
Molecular Formula:
C59H97ClN2O4
Molecular Weight:
933.88
CAS Number:
41085-99-8
Protocol (PDF):
C017
MSDS (PDF):
MSDS-C017
MSDS (PDF):
C017
Mol Biol Cell (2001) 12:919-930
J Immunol Methods (2006) 308:53-67
Immunol Cell Biol (1999) 77:499-508
Lipophilic cyanine dyes, such as DiO (Cat. No. C016), DiI (Cat. No. C017), DiD (Cat. No. C018) or DiR (Cat. No. C019), are commonly used. The longer the alkyl chain on the dye, the better the retention in lipophilic environments.
This is expected. DiD (which is far-red fluorescent) is never as uniform as DiI (which is orange fluorescent). If uniformity is desired, try increasing the label time and concentration, but it still isn’t likely to be as uniform as DiI.
A typical method is to label one cell line with orange fluorescent DiI and the other cell line with green fluorescent DiO. These orange and green lipophilic cyanine dyes will stain the membranes of cells. Cells that fuse will then have both dyes, yielding a yellow color (when images are overlaid or cells are imaged in a dual-bandpass filter). These live cells can then be labeled with Hoechst 33342 (a cell-permeant blue DNA stain comparable in wavelength to DAPI), but only as an endpoint just before imaging (since DNA stains can interrupt DNA function).
Home » Cell Structure Probes » DiI perchlorate
DiI perchlorate
Introduction
To Order
Documents
Cocco RE, Ucker DS
Gómez-Román VR, Florese RH, Patterson LJ, Peng B, Venzon D, Aldrich K, Robert-Guroff M
Parish CR
Contact Us
ABP Biosciences
405 E Gude Dr, STE 214
Rockville, MD 20850
Service Hotline
Tel: 301-658-7993
E-mail: info@abpbio.com
Copyright@ 2026 ABP Biosciences, LLC. All Rights Reserved.

